Nature’s Blueprint: Plants Did It First

Billions of years before humanity dreamed of green hydrogen, plants were already splitting water with sunlight. Are we finally reading the manual?
eyesonuruguay
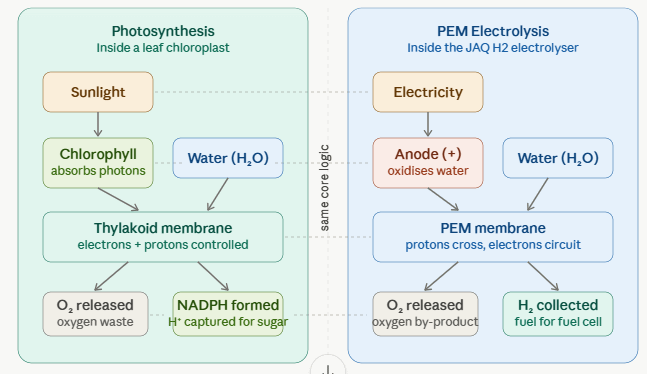
Inside every leaf on Earth, a silent revolution happens trillions of times per second. Water molecules are torn apart. Hydrogen is freed. Energy is stored. It is the most fundamental chemistry on our planet — and engineers building the JAQ H1’s next-generation electrolyser are, whether they know it or not, reinventing a wheel that evolution perfected 2.7 billion years ago.
The connection is not merely poetic. When you strip both systems to their essentials, plant photosynthesis and industrial electrolysis are solving the exact same problem — how to split H₂O into its parts using an energy input — and they arrive at strikingly similar solutions. The story of how humanity is learning, consciously and sometimes unconsciously, from nature’s oldest energy technology is one of the most compelling threads in modern science.
The core reaction: tearing water apart
Water (H₂O) is a stubbornly stable molecule. The bond between its hydrogen and oxygen atoms doesn’t break easily; you need to inject energy. Both plants and electrolysers do exactly this, just with different energy sources and different molecular machinery.

The output in both cases? Hydrogen and oxygen, separated. In the plant, the hydrogen doesn’t stay as H₂ gas — it’s immediately seized by a molecule called NADP⁺ and used to build sugar. In the electrolyser, the hydrogen is collected as pure H₂ gas, ready to power a fuel cell. The destination differs; the starting move is identical.
The electron transport chain meets the PEM membrane
The real elegance — and the real parallel — lies not just in what’s split, but in how the splitting is managed. In both systems, the process hinges on carefully controlling the movement of electrons.
In a leaf, after chlorophyll absorbs light, freed electrons travel through a series of proteins embedded in the thylakoid membrane — the electron transport chain. As they flow “downhill” energetically, they pump hydrogen ions across the membrane, creating a concentration gradient that powers the production of ATP, the cell’s energy currency. It is an extraordinarily elegant nanoscale power station, and it has been running at near-perfect quantum efficiency for billions of years.
In a modern PEM (Proton Exchange Membrane) electrolyser of the type planned for the JAQ H2 vessel, a polymer membrane separates the two electrodes. Water is fed to the anode; the electrical current strips electrons from the water molecules and sends hydrogen ions — protons — through the membrane to the cathode, where they combine to form H₂. The membrane is the gatekeeper, just as the thylakoid membrane is in the chloroplast.
At the molecular level, the thylakoid membrane and the PEM electrolyser membrane are performing the same trick: allowing protons through while keeping electrons on a separate controlled path.
Side by side: the parallels
| Function | Photosynthesis (plant) | Electrolysis (JAQ H2) |
|---|---|---|
| Energy input | Sunlight (photons) | Electricity (from renewables) |
| Raw material | Water (H₂O) | Water (H₂O) |
| Water splitting | Photolysis at Photosystem II | Oxidation at the anode electrode |
| Electron manager | Thylakoid membrane + electron transport chain | PEM membrane + electrode circuit |
| Proton shuttle | H⁺ ions cross the thylakoid space | H⁺ ions cross the PEM membrane |
| Hydrogen output | Captured by NADPH (for sugar synthesis) | Released as H₂ gas (for fuel cells) |
| Oxygen output | Released as O₂ (originally toxic waste!) | Released as O₂ (harmless by-product) |
| Energy storage format | Chemical: glucose (C₆H₁₂O₆) | Chemical: hydrogen gas (H₂) |
Are we learning from nature? The “artificial leaf”
The short answer is: yes, increasingly and deliberately. The field of artificial photosynthesis has been explicitly attempting to replicate the plant’s trick for decades. The landmark moment came in 2011 when MIT chemist Daniel Nocera unveiled what he called the “artificial leaf” — a silicon chip coated in catalysts that, when dropped in water and placed in sunlight, split water into hydrogen and oxygen, mimicking photosystem II.
Research milestone (2025)
A team at UNIST (South Korea) published a modular artificial leaf in Nature Communications (2025) that produces hydrogen from sunlight and water without any external power source — achieving solar-to-hydrogen conversion efficiencies that surpass a key 5% commercial benchmark. Researchers at JAIST and the University of Tokyo have simultaneously developed bioinspired hydrogels that split water using sunlight, showing that polymer networks can replicate the energy conversion role of chloroplasts.
Meanwhile, research at DGIST in South Korea has gone even further — using bacteria with hydrogenase enzymes (the very enzymes plants use to handle hydrogen) combined with light-harvesting dyes, building a solar biohydrogen system that explicitly fuses the biological and the synthetic. Some solar cell-based water-splitting systems are now reported to be approximately ten times more efficient than natural photosynthesis at converting sunlight to hydrogen — a humbling fact for evolution, but a promising one for engineering.
Where nature still wins
For all our ingenuity, plants hold several cards that engineers haven’t yet matched. Natural photosystems have a quantum efficiency that approaches 100% for the initial electron transfer step — almost every absorbed photon triggers a reaction. They are self-repairing: damaged proteins in the electron transport chain are automatically recycled and replaced. And they have been optimised by 2.7 billion years of evolutionary pressure to work at ambient temperature, in seawater, at low cost, using only Earth-abundant elements.
Current PEM electrolysers often rely on platinum or iridium as catalysts — rare, expensive metals. Plants use manganese and iron. Researchers studying the oxygen-evolving complex of Photosystem II — essentially nature’s water-splitting catalyst — are trying to replicate this manganese cluster in synthetic materials, with promising early results. In 2024, a team reported a manganese-iridium hybrid catalyst as a “quantum leap” toward precious-metal-free hydrogen production.
Every advance in green hydrogen technology is, at its core, an attempt to write a shorter version of a 2.7-billion-year-old instruction manual.
What this means for the JAQ H2
The JAQ H2 vessel’s planned onboard electrolyser — feeding both itself and the JAQ H1 with green hydrogen drawn from the seawater around them — is not nature-mimicking in the deliberate sense of artificial photosynthesis research. It uses conventional PEM technology powered by renewable electricity, not sunlight-to-hydrogen direct conversion. But the conceptual kinship is undeniable: a vessel floating on the ocean, using the water around it as both raw material and fuel source, producing only oxygen as a waste product.
That is, in essence, what a leaf does on a sunny afternoon. And when the JAQ H2 glides through the Amazon basin, surrounded by the world’s largest living collection of chloroplasts, it will be doing something those leaves have done since before the dinosaurs — just with different machinery, and for different passengers.
Nature invented this technology first. We are only now beginning to understand the instruction manual.

